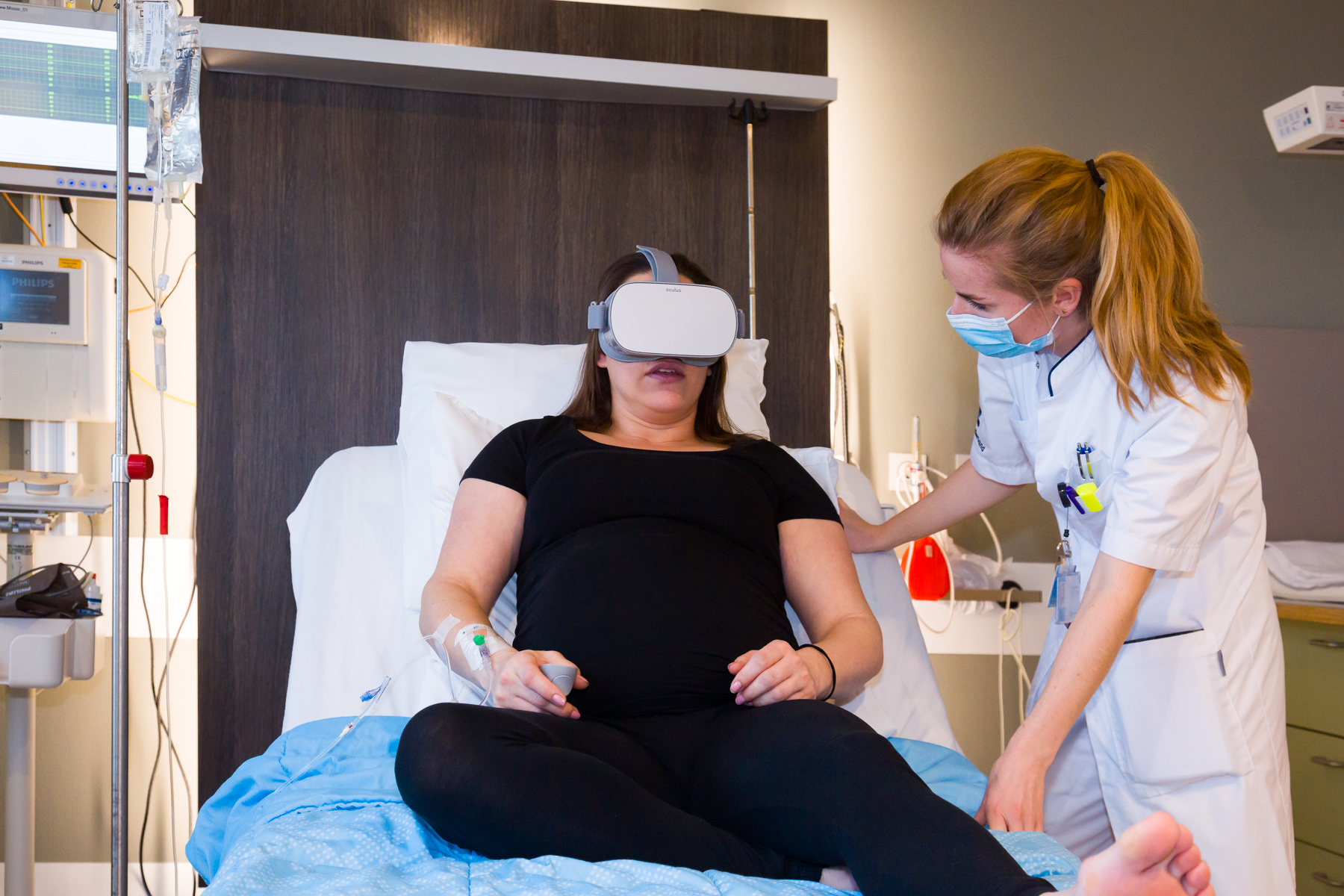
There is increasing evidence that virtual reality (VR) is effective in the reduction of labour pain. The implementation of alternative methods like VR to reduce labour pain can contribute to reduce patient request for pharmacological pain management methods and associated side effects. The aim of this study is to examine women’s experiences, preferences and satisfaction in regard to the use of VR during labour.
Methods
A qualitative interview study was conducted in a non-university teaching hospital in The Netherlands. Two VR applications, respectively a guided meditation and an interactive game were tested in eligible women with a sin- gleton pregnancy, scheduled for induction of labour. For the primary outcome, patients’VR experience and applica- tion preference (meditation vs. game) were examined using a post-intervention questionnaire and a semi-structured interview. Three categories (with sub-categories) were used to guide interviews: “The VR experience”, “Pain reduction”, and “Usability of the VR application”. Labour pain before and directly after VR was evaluated using the NRS score.
Results
Twenty-four women, of whom 14 were nulliparous and ten multiparous, were included and 12 of these women participated in semi-structured interviews. Using within-subject paired t-test comparisons, compared to pain pre-VR, patients reported a highly significant 26% decrease in mean NRS scores during VR meditation (pain pre-VR = 6.71 + − 1.65 vs. pain after VR = 4.96 + − 2.01) [p < 0.001]. Compared to pain before VR game, patients also reported a highly significant 19% decrease in mean NRS scores during VR game (pain before VR game = 6.89 + − 1.88 vs. pain after VR game = 5.61 + − 2.23) [p = 0.001].
Conclusion
All women were highly satisfied with VR use during labour. Patients reported a highly significant reduc- tion in pain during the interactive VR game and during meditation, patients preferred guided meditation. These results can contribute to the development of a potential promising new non-pharmacological tool to reduce labour pain.
Trial registration ClinicalTrials.gov Identifier: NCT04858984
Bron: Musters et al. BMC Pregnancy and Childbirth (2023) 23:283
Andere artikelen

‘Dit heeft me echt geholpen’
Ik vond het heel fijn om echt iets praktisch te kunnen doen tijdens mijn zwangerschap. Ik had geen ervaring met mediteren ...

Bevallen met Virtual Reality
Bron: Nederland Tijdschrift Obstetrie en Gynaecologie. Virtual Reality (VR) is een virtuele werkelijkheid die ontstaat doo...

Waarom mediteren?
Bevallen met BirthCoach VR? Lees hier meer over het effect van meditatie. Meditatie is een techniek die zorgt voor rust, o...

‘Een perfecte uitkomst’
“Na het afzetten van de VR bril bij de laatste keer gebruik kreeg ik persweeën en werd heel snel daarna onze zoon g...